Abstract
Contents
- 1 Abstract
- 2 Introduction
- 3 Materials and methods
- 4 Results
- 5 Discussion
- 6 Conclusion
- 7 Data availability
- 8 References
- 9 Funding
- 10 Author information
- 11 Ethics declarations
- 12 Additional information
- 13 Rights and permissions
- 14 About this article
- 15 Comments
The IEEE and ICNIRP have set the maximum permissible exposure for an instantaneous peak electric field of 100 kV/m. However, no reason was given for this limitation. A new exposure system is designed through a detailed process of analytical analysis, numerical modeling and prototype testing. The system consists of a cylindrical re-entrant resonant cavity which can reach an electric field strength of more than 100 kV/m with an input power of 200 W. The work of the system was evaluated in simulations and experiments in terms of scattering parameters, electric field distribution and specific absorption rate. The system was then used to perform in-vitro exposure of the human lymphoid cell line (GG0257) to a 1195 MHz signal at a peak power of 53 dBm and a pulse width of 550 ns over a range of interpulse intervals to identify heating-induced changes in cell viability. The proposed system offers a high Q value of 5920 under no-load conditions which is lowered to 57 when loaded with 12 ml of cell culture but still offers a field intensity of 67 kV/m. Using the system for exposure of GG0257 cells lasting for 18 min, interpulse intervals of 11 s or less led to a reduction in the number of viable cells and an increase in the corresponding necrotic cells. For a shorter exposure duration of 6 min, a decrease in cell viability was seen at interpulse intervals of 5.5 s or less. The designed exposure system is capable of handling high-intensity electric fields well. Temperature measurements with fiber optic probes and temperature sensitive labels showed that changes in viability were associated with an increase in temperature above 46 °C. This novel exposure system is an efficient way to investigate the possible relationship between peak field intensity and biological effects to provide the rationale behind the 100 kV/m maximum exposure limit.
Introduction
Science-based guidelines for exposure to electromagnetic (EM) fields, such as those published by ICNIRP1 and IEEE2, are based on the well-established effects of these fields on biological systems. For the radiofrequency EM field, the defined biological effect is tissue heating and these guidelines are intended to limit the temperature rise caused by time-averaged heating3.
Previous versions of the two guidelines set a peak field intensity limit for pulses. The IEEE 20052 standard specifies the maximum permissible exposure to an instantaneous peak electric field of 100 kV/m. No reason is given in the 2005 standard for this limit, but earlier versions of standard4 state that:
“The recommendation for a peak E field limit of 100 kV/m is based on the need to limit the permissible field below the level at which air damage or spark release occurs.”
Similarly, the 1998 ICNIRP guidelines suggest that, for frequencies exceeding 10 MHz, the equivalent field wave power density averaged over the pulse width should not exceed 1000 times the reference level or that the field strength should not exceed 32 times the reference field strength level5, whereas acknowledge that:
“Little information is available about the relationship between biological effects and pulse field peak values.”
Thus, although limits were defined for peak field intensities, these were not based on known biological effects and were removed from the most recent version1.
Although various low-power exposure systems for studying the biological effects of EM fields emitted from microwave equipment have been described in the literature6,7,8,9,10,11,12,13,14,15,16,17,18, systems that can handling very high electric fields is relatively rare. In addition, these exposure studies have struggled with issues of re-creation, reproducibility, and reliability19. In-vitro exposure studies also require uniform field distribution to ensure that all cells are exposed to a similar Specific Absorption Rate (SAR). A further requirement is for the system to fit inside the cell culture incubator.
Classic closed exposure systems capable of providing high electric fields can be divided into three categories19; (1) TEM cell exposure system; (2) rectangular waveguide exposure system; and (3) resonant cavity exposure system. A typical TEM cell is a rectangular coaxial transmission line tapering at each end forming a metal shielded environment that establishes the Transverse Electro-Magnetic (TEM) mode of the EM field over a certain frequency range. This type of TEM cell exposure system has long been used in exposure studies since its discovery by Crawford7,11,20,21,22,23. TEM cells provide the exposure conditions closest to the free space environment. Although, the electric and magnetic fields are well characterized, maintaining the temperature in a TEM cell is not easy and a thermal gradient is always present. Therefore, they are not the preferred choice for high power exposures.
Rectangular waveguides are another popular category of exposure systems. They limit the power inside the equipment due to the total reflection of EM waves from the waveguide walls16,24,25,26,27,28,29,30. However, these systems are bulky and difficult to incorporate into cell culture incubators, making environmental control difficult. In addition, they have a narrow operating frequency range and the limited interior size of the waveguide exposure chamber limits their use for certain types of cell culture containers19.
The resonant cavity exposure system has also been used extensively in the study of biological effects on cell culture. They offer high volume efficiency and compact size, allowing for easy placement in the incubator19. The electrical parameters of resonant frequency, quality factor (Q) and impedance matching are very sensitive and, therefore, very difficult to achieve11. A suitable resonant structure absorbs all the incoming power increases efficiency and has the capability of high electric field magnitude31. A number of resonance systems for exposing cell cultures have been presented in the literature. Although, most of these resonant cavity exposure systems use a rectangular waveguide shortened at one end32,33,34,35,36,37,38, the cylindrical structured resonance system can irradiate a closed sample with a very uniform field throughout the entire crucible39, 40 ,41,42,43. The cylindrical resonance cavity has also been designed for in vivo dental dosimetry44, homogeneous hydrolysis of the enzyme sucrose45, measurement of general solvent dielectric46, and electron paramagnetic resonance spectroscopy47. Re-entrant cavity resonators further enhance these characteristics and have been used in many applications including particle accelerators, electron spin spectroscopy, dielectric characterization, microwave oscillators, displacement sensors, tunable resonators, and filters43,48.
As ICNIRP acknowledges, there has been little research on a possible relationship between peak field intensity and biological effects. This study is an attempt to address this important issue. The literature indicates that TEM cells are not favored for high power exposures, whereas rectangular waveguides are not suitable for the purposes of this study due to size and power handling constraints49 introduced by the available hardware (Agilent E4432B signal generator and incubator). The resonant cavity appears to be a good instrument for studying this effect offering a simple geometry, high Q level, and a uniform field pattern throughout the sample container.
This work has developed a new system based on a cylindrical re-entrant resonant cavity that can achieve high electric field strengths for cell culture exposure. The system supports field strengths in excess of 100 kV/m with an input power of 200 W. Under no-load conditions, the system exhibits good impedance matching at 1800 MHz, with a Q factor of 5920. Load cavities with a 55 mm diameter petri dish containing the cultured cells. shifted the resonant frequency to 1195 MHz and reduced the Q factor to 57. A detailed numerical analysis followed by reflection coefficient measurements was performed to validate the working of the exposure system. The human lymphoid cell line (GG0257) was then used to identify exposure thresholds for heating-induced biological effects using the designed exposure system, which was a necessary requirement before looking for peak field effects only (i.e. no heating). The duty cycle is reduced (by increasing the interpulse interval) while maintaining a constant peak field, until the energy absorbed is insufficient to cause the heating-induced decrease in cell viability. Future experiments will use duty cycles below this threshold to look for effects that may be specifically related to peak field intensity. The results of this work will provide evidence for assessing peak field boundary requirements in exposure guidelines for pulses.
Materials and methods
The cylindrical re-entry cavity structure can be used to generate electric fields in different modes. The re-entrant cavity consists of a short metal section with a gap in the center conductor. Re-entrant refers to the extension of the metal boundary into the interior of the cavity, as shown in Fig. 1.
The re-entrant structure limits the electric field in a small area which increases its strength significantly. It also maintains a high Q factor ((sim )3000) because the magnitude of the magnetic field is small and spread over a much larger volume resulting in low surface losses. Thus, the re-entrant cavity has the advantages of simple mechanical construction and tuning capability over a wide frequency range while having a small overall size. In addition, these cavities are highly sensitive to detect even small changes in field strength and offer a fairly uniform electric field distribution in the exposed sample. They are widely used in klystron, solid-state devices and dielectric measurements and these advantages led us to consider them for exposure of cells to very high RF fields (i.e. strengths >100 kV/m) in this study.
A cylindrical re-entrant cavity resonator with major structural dimensions; (a) Cavity structure; (b) Key dimensions and field configuration for mode (TE_{111}) (traverse: H-plane in, circle: H-plane out, arrows: E-plane).
Analytical design
Key parameters that determine re-entrant cavity performance include cavity length (( l )), distance between re-entrant stage and cavity wall (( d )), re-entrant stage radius (( a )) and cylinder radius (( b )). Different techniques can be used to feed the cavity whose loop structure is the most adopted.
The basic mode ((TE_{111})) radial cavity frequency shown in Fig. 1 can be calculated using the following expression48,50:
$$begin{aligned} f= & {} frac{c}{2 pi sqrt{epsilon _r} } left[ al left( frac{a}{2d} – frac{2}{l} ln {frac{0.765 }{sqrt{ l^{2} + {left( ba right) }^2 } } } right) ln frac{b}{a} right] ^{-frac{1}{ 2} } end{harmony}$$
where (c = 3 times 10^8) m/s is the speed of light and (epsilon _r) is 1 for an air-filled cavity. There can be an infinite number of modes in such a cavity. The field distribution for the (TE_{111}) mode is illustrated in Fig. 1b. In this mode, the electric field is concentrated in the gap region which makes high electric field strengths achievable. Therefore, the (TE_{111}) mode was chosen as the dominant mode in the proposed cavity design.
The field strength inside the resonator cavity depends on the factor Q which is calculated as 51,52,53:
$$begin{aligned} {Q}_c=frac{ {2{omega }_o} {W}_e}{{P}_c} end{aligned}$$
where, (W_{e}) is the stored electrical energy while ({P}_c) is the power dissipated in the cavity walls. The operating frequency is given by ({omega }_o).
Numerical modelling
The design of the in-vitro exposure system should follow the set of requirements utilized by the available test equipment. These requirements are as follows:
It should reach a peak electric field intensity of 100 kV/m with an input power of 200 W.
The resonant frequency under no-load and load conditions must be between 1000 and 2000 MHz, limits imposed by the available power source (Agilent E4432B signal generator).
The system must be compact with the largest dimensions of no more than 150 mm to fit the incubator.
System dimensions must be large enough to accommodate 55 mm petri dishes containing cell cultures while providing a stable platform for the sample and uniform distribution of the field throughout the container.
The numerical design is carried out through a painstaking optimization process because the individual dimensions for the required performance cannot be calculated directly. Equation (1) governs the choice of the initial cavity dimensions. CST Studio Suite software using Finite Integration Technique (FIT)54 was used to model the proposed exposure system and analyze its performance numerically. A paired loop structure is used to excite the (TE_{111}) mode in the cavity because it has a relatively wider range of reactive matching. The reflection coefficient, electric field and Q factor were used as performance metrics.
Unloaded cavity
A numerical model of the initial design of the proposed cylindrical re-entrant cavity along with the structural dimensions is given in Fig. 2. Power is fed to the system using an N-type coaxial connector. A PTFE collar with a dielectric constant of 2.1 was inserted to hold the petri dish and stabilize the cell culture. To increase the field strength in the cavity and improve mechanical stability, a double reentry arrangement is used. The gap between the two re-entry stages is kept at 16.2 mm to provide sufficient space for the petri dish with the lid.
The schematic layout of the initial design of the proposed cylindrical re-entry cavity exposure system (all lengths in mm) shows a cross-sectional view of the cavity loaded with 12 ml cell cultures contained in a petri dish.
The performance of the proposed exposure system is first analyzed in terms of the reflection coefficient ((S_{11})) response shown in Fig. 3a. Strong resonance was observed at 1350 MHz when the cavity was lowered. The magnitude of the reflection coefficient was recorded at 16 dB which indicates a very good fit. Figure 3b presents the electric field distribution seen on the center surface of the cavity calculated on 10 curves covering the entire region of the gap between the two re-entry stages.
Analysis of the performance of the proposed exposure system; (a) Reflection coefficient response under no-load and load conditions; (b) Electric field profile on curves covering the length and width of the cavity under unloaded conditions.
The electric field distribution shows that the system can reach a peak field strength of 120 kV/m. Also, the plane profile remains largely uniform throughout the cavity. The high peak indicates the metallic edge of the larger re-entry stage. The Q factor for the proposed system was recorded as 9533. These results indicate that the design of the cylindrical re-entrant resonant cavity exposure system efficiently satisfies all the design requirements.
Loaded cavity
The resonant cavity is intended to be used to expose human tissue cells to very high electric fields making it relevant to evaluate their performance under loading conditions. Standard polystyrene petri dishes 55 mm in diameter, 14.2 mm high and (epsilon _{r} )=2.4 were designed to accommodate cell cultures for exposure studies55,56. These petri dishes are an optimized choice supported by the size of the incubator and the limited space available in the cavity chamber. Using a larger petri dish will require a larger cavity and affect its ability to fit into the available incubator while a smaller petri dish cannot be mechanically stabilized. The use of more than one petri dish is no longer feasible due to the form factor of the system. Moreover, it will have performance implications with respect to reflection response and electric field distribution profiles.
Cell culture medium 12 ml (RPMI 1640, Roswell Park Memorial Institute Medium) with a relative permittivity, (epsilon _{r} )=66.14 and a conductivity, (sigma )=1.81 S/m ( based on measurements at 1275 MHz and 37 °C) having a mass density, (rho )=1050 kg/m(^3) contained in a 55-mm petri dish was considered to simulate and replicate the worst case loading scenario.
The geometry of the filled cavity is illustrated in Fig. 2. The results of the reflection coefficients are given in Fig. 3a shows that loading cavities with 12 ml cell cultures in petri dishes greatly affects performance by including impedance mismatch and resonance shift to 883 MHz.
Optimisation of exposure system
Since loading the exposure system with cell culture shifts its resonant frequency out of the desired band (ie 1000–2000 MHz), the cavity design needs to be optimized. When loading cavities with petri dishes and cell culture introduces a capacitive effect, the inductive loading of the system can effectively balance it and minimize the frequency drop.
Resets the loaded cavity exposure system by changing the coupling loop dimensions.
Two balancing techniques are applied to realign the filled voids in the desired band; (1) various dimensions of the coupling loop; and (2) reducing the free space in the cavity. Varying the coupling loop dimensions can change the impedance matching significantly. Different variations in the loop structure were investigated in the simulation to see the effect on the reflection coefficient. The three parameters that vary are the radius, length and width of the coupling loop. The reflection coefficient curves for these variations are compared in Figure 4. A significant effect of changing the coupling loop dimensions on impedance matching can be observed. Although the third dimension of the loop structural parameters affects the impedance matching, adopting a single parameter that is mostly changing will not work due to the limited space in the cavity. Therefore, all three parameters need to have carefully optimized enhancements for better impedance matching. Therefore, loops with dimensions of length 30 mm, width 15 mm and radius 2 mm were adopted based on this optimization study.
The second method involves reducing the space in the cavity. The metal blocks, 2 to 4 numbered and placed at different angles to the vertical axis of the cavity, are considered as shown in Fig. 5a-d. Figure 5e compares the performance of the cavity reflection coefficient with varying beam counts and is excited using a larger coupling loop. The results show that the inclusion of a metal block does improve impedance matching and cavity realignment via an upward resonant shift. The larger the number of blocks, the greater the frequency shift with the inclusion of four blocks giving the best performance. Since the four beams touch each other at the front end, it was hoped that a horseshoe-shaped semicircular metal ring would work better.
The geometry of the re-entrant cavity is a cylindrical loaded re-entrant with a metal block; (a) Two blocks; (b) Two beams at 45°; (c) Three blocks; (d) Four blocks; (e) Reflection coefficient response for cavities loaded with metal beams.
Optimised unloaded cavity
The impedance matching technique study revealed that the loaded cavity performs well when a larger excitation loop together with a semicircular metal ring for inductive loading is used. Based on these findings, the design of the cylindrical cavity resonator was updated. Numerical models are simulated and the results are analyzed.
Although, the larger excitation loop works well for the loaded cavity, it fails to provide the same impedance matching for the unloaded scenario. Therefore, the N-type connector is designed to support switchable coupling loops. The original smaller loop was used for the cavity that was lowered and needed to be removed and replaced with a larger loop when the cavity was used for cell culture exposure. Since, the main use of the cavity will be in the loaded state, loop replacement is rarely required and mostly for confirmation of simulation results through experiments.
Instead of inserting four separate metal blocks, a horseshoe-shaped semicircular metal ring is designed as an inductive load. The modified cavity design is presented in Fig. 6a. Due to this addition, a slight change to the smaller coupling loop was required. fig. 6b, c illustrate the optimized geometry of the smaller and larger coupling loops. The performance of the modified disassembled cavity is then analyzed in the simulation. The reflection coefficient response is given in Fig. 3a whereas the electric field distribution is calculated on a 2 mm apart curve covering the length and width of the gap region between the two cavity re-entry stages is shown in Figure 7. The results show that the modified resonant cavity exhibits excellent impedance matching with strong resonance at 1800 MHz. The electric field distribution shows that with the introduction of the semicircular metal ring, the electric field strength has increased to 125 kV/m due to the reduced non-metallic area with a uniform pattern in the central region where the cell culture will be placed. The peak that appears on the lower Y axis is due to the proximity of the coupling loops. The Q factor has seen a decrease to 5920 indicating increased losses due to the higher volume of the metal surface.
Optimized cylindrical re-entrant re-entrant resonant cavity exposure system geometry with semicircular metal rings for impedance matching (all lengths in mm); (a) Cavity components; (b) Smaller loop dimensions for unloaded cavity excitation; (c) Larger loop dimensions for loaded cavity excitation.
Electric field distribution for optimized re-entrant resonant cavity exposure system with semicircular metal ring; (a) Disassembled cavities; (b) The cavity is filled with 12 ml of cell culture.
Optimised loaded cavity
A modified cylindrical resonant cavity with a larger coupling loop for excitation was then tested under loading conditions. The cavity was filled with a 55 mm petri dish containing 12 ml of cell culture medium. Petri dish dimensions and electrical properties of cell cultures have been discussed previously.
The simulated reflection coefficient (Fig. 3a) shows that excellent impedance matching is achieved even though the petri dishes and cell cultures are loaded. Although, the resonant frequency is shifted to 1195 MHz, it is still well above the lower end of the desired band (i.e. 1000 MHz). It is clear from Figure 7b that, although the electric field strength is reduced due to energy deposition in the cell culture, the degree of reduction is smaller compared to the original loaded cavity design. A maximum field strength of 63 kV/m and a Q factor of 57 were noted for the optimized cavity design.
The results show that the optimized cylindrical re-entrant resonant cavity exposure system meets all design requirements. It achieves a peak electric field strength of over 100 kV/m under no-load conditions. In addition, it efficiently overcomes the problem of impedance mismatch and resonance shift beyond the desired frequency.
Numerical exposure study
The optimized cavity model was used for detailed numerical exposure studies. The effect of various cell culture volumes in petri dishes on impedance matching, resonant frequency, electric field strength and SAR distribution was studied through computer simulation. The input power is considered to be 200 W and the cell culture volume varies from 2 to 22 ml.
Performance evaluation of exposure systems designed with varying cell culture volumes; (a) Reflection coefficient response; (b) Electric field strength; (c) SAR (mean value and Coefficient of Variation).
Distribution of SAR for various cell culture volume exposures in the horizontal plane in the center of the cell culture; (a) 2 ml; (b) 4.5 ml; (c) 7ml; (d) 9.5 ml; (e) 12 ml; (f) 14.5 ml; (g) 17ml; (h) 19.5 ml; (i) 22ml.
Figure 8a comparing the simulated (S_{11}) responses for filled cavities shows that an increase in the cell culture volume causes a decrease in the resonant frequency in the 50–60 MHz interval. For 2 ml of cell culture, the resonance frequency is 1435 MHz to 954 MHz for 22 ml of cell culture. It was therefore concluded that the maximum volume should not exceed 19.5 ml to maintain resonance within the operating bandwidth of the power amplifier. The best impedance matching of 22 dB was observed for a 9.5 ml cell culture volume with a resonant frequency of 1264 MHz.
The electric field is calculated on the vertical center axis of the cavity and plotted in Figure 8b which provides insight into the formation of the field in the presence of cell cultures. It can be observed that increasing the cell culture volume decreases the electric field strength due to higher adsorption. The minimum field strength was observed to be 1.55 kV/m for 22 ml of cell culture.
Exposures were characterized for different cell culture volumes in terms of mean SAR value (calculated using IEEE standard C95.357) and Coefficient of Variation (CV). CV is a measure of the non-uniformity of the SAR56 value. Results in Fig. 8c reflects the strong dependence of exposure on cell culture volume. The cell culture volume of 12 ml gave the best performance with a CV of 30% and an average SAR value of 4300 W/kg. Although the 14.5 ml cell culture produced the least amount of non-uniformity, it had a lower mean SAR of 3867 W/kg.
The distribution of SAR for cell culture exposure in a horizontal section through the center of the cell culture medium (Fig. 9) indicates that the SAR level is highest near the surface of the petri dish illuminated by the coupling loop. Then tapering towards the other end in a uniform circular ring having a minima in the center of the petri dish. Higher cell culture volumes offer relatively better uniformity.
Fabrication and testing of exposure system
The optimized design of the cylindrical re-entry cavity resonator was fabricated at the Engine Workshop and tested at the Antenna Measurement Laboratory at Queen Mary University of London. The cavity walls are made of aluminum and contain a PTFE collar. Brass is used for the two clutch loops. A 50 ohm N type connector is included for the excitation signal port. The artificial exposure system is shown in Figure 10a. In order to reduce the effects of corrosion and improve performance, the fabrication cavities were gold-coated through gold plating with an effective gold layer thickness of 25 m (Fig. 10a).
Cavity performance was analyzed both under unloaded and loaded conditions. Since, saline solutions play an important role in many chemical and biological systems, they are useful tools for establishing the workings of biological exposure systems. Therefore, the operation of the artificial cavity under loading conditions was tested by measuring the reflection coefficient carried out using a saline solution (224.5 g/L sodium chloride solution supplied by APC Pure and deionized water). Petri dishes contained 12 ml of saline solution, similar to the simulated cell culture exposure conditions. The cavity is excited using a larger coupling loop under load conditions.
Testing the prototype made; (a) Artificial exposure systems; (b) Experimental setting.
Reflection coefficient measurements
The created cavity was first tested by measuring the reflection coefficient ((S_{11})) in different configurations to confirm the simulation results. The two-port VNA (Hewlett Packard 8720ES-VNA) shown in Figure 10b was used for measurements.
The comparison of the measured reflection coefficients for the non-coated (aluminum) and gold-coated cavities under no-load conditions with the simulated response is illustrated in Figure 11. The three results are very well matched. It was observed that gold plating has improved performance, made the reflection coefficient stable and removed ripples. Simulation results and measurements of the gold-plated cavities that were disassembled and filled with petri dishes containing saline solution also showed a very good fit. The disassembled cavity operates fine at 1800 MHz in simulation and 1790 MHz in measurement. The loaded cavity also performs well in terms of resisting detuning caused by saline solution replicating cell cultures. The recorded resonant frequency is 1325 MHz in the simulation versus 1360 MHz in the measurements, close to the 1500 MHz center frequency in the desired 1000-2000 MHz frequency band.
The reflection coefficient measurements from the exposure system compared the simulated and measured reflection coefficients for unloaded and loaded non-coated and gold-plated cavities (with 12 ml of cell culture).
Power leakage test
Since the exposure system has very high input power levels, power leakage can cause severe damage to the power amplifier, incubator, and other modules. To ensure system safety, the proposed cavity is tested for electromagnetic leakage. A signal generator, spectrum analyzer and dipole probe are used for this purpose. A 25 dBm CW (continuous wave) generated by the signal generator is fed to the cavity. The probe is used to monitor any spectrum changes at 1790 MHz to observe power leakage at different positions, especially near the feeding point. Cavities are placed inside the anechoic chamber to reduce electromagnetic interference.
Measurement of the power leakage test of the gold plated exposure system by observing the power spectral density at various points in the cavity.
The response spectrum presented in Fig. 12 shows that the probe picks up a high signal at 1790 MHz near the excitation loop for the open cavity. However, for closed cavities, no signal above the noise floor is recorded. The strongest signal in this configuration is at the 56 dBm level. This clearly shows that the exposure system does not have a significant level of power leakage. Therefore, an exposure system was created, ready for biological studies.
Ethics approval
This study was conducted in accordance with the International Ethical Guidelines for Epidemiological Studies.
Results
This study was excluded from the application of these guidelines because no human subjects were used in the study
Experimental exposure setup
investigation.
Cell dosimetry
An RF exposure system based around a resonant cavity has been simulated, prototyped, and tested. The results show that the system achieves the required set of specifications for high pulsed field exposure at electric fields >100 kV/m. This section details the experimental use of the system to study cell culture exposure.
Cell viability study
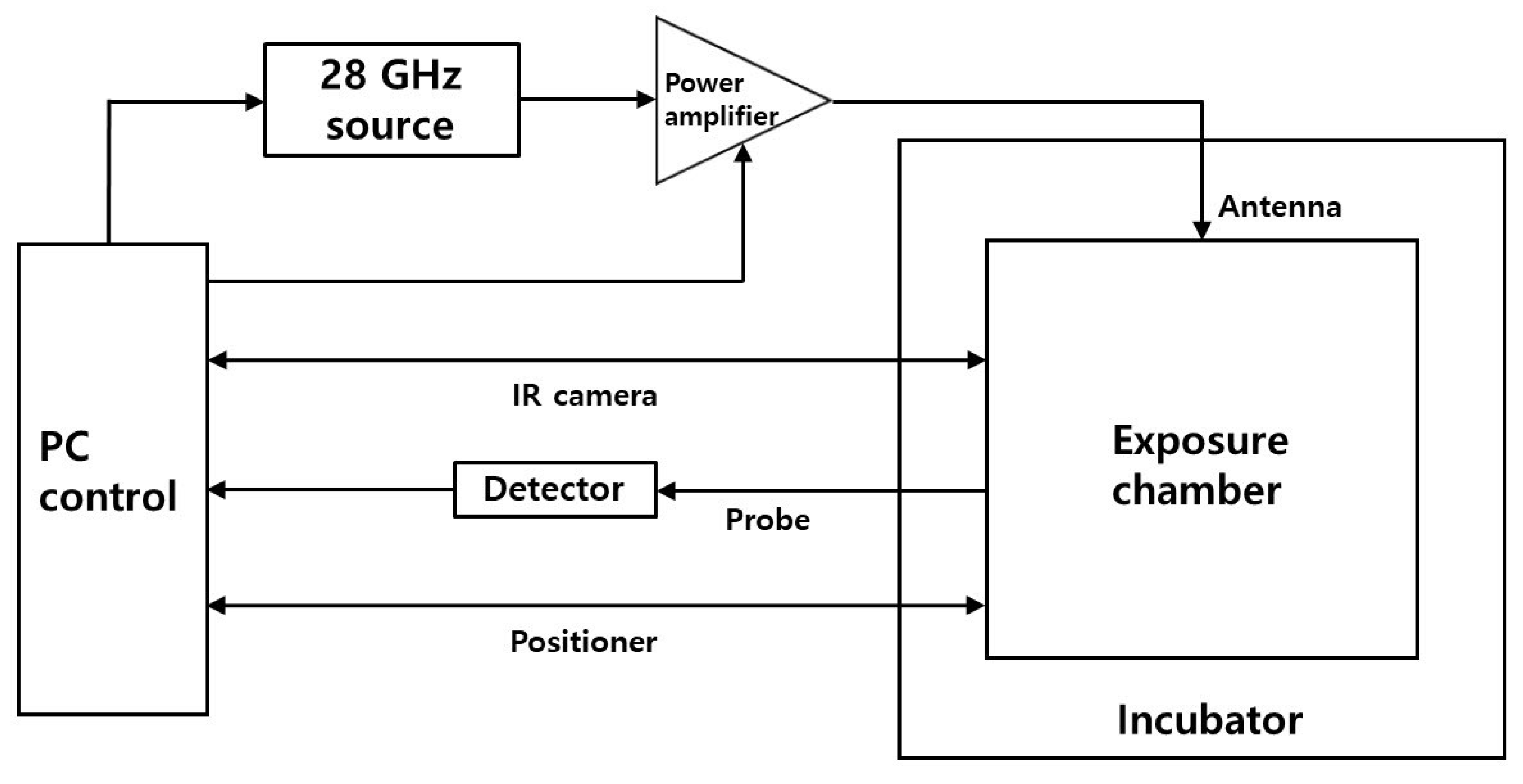
The resonant cavity is driven by 200 W (53 dBm) of RF energy. The signal is generated by the Agilent E4432B signal generator and divided using a 3 dB splitter. Half of it is monitored by the Tektronix DPO7254 oscilloscope, and the second half is fed via the Research Amplifier Research (AR) 250W1000C power amplifier to the input of the Varian VZL-6943G5 TWT amplifier. The output of the TWT amplifier is then fed through the DC7144A Amplifier Research directional coupler and the Succofeed transmission line to the resonance cavity. The directional coupler protects the power amplifier’s output stage from reflection and also allows the Agilent E4417A power meter to monitor forward and reverse power between the amplifier and the resonant bay. Pulses are provided by an Agilent 8118A pulse generator, the output of which is monitored by an oscilloscope and fed to a signal generator to control the output signal generator. A resonant cavity was placed inside the incubator to control the temperature at 37 °C. Fixed exposure conditions included a 1195 MHz signal at 53 dBm peak power with a pulse width of 550 nS. The exposure duration and duty cycle (interpulse interval) are changed to control the exposure level.
Temperature measurements
GG0257 cells (European Cell Culture Collection, Public Health England, UK) were cultured in RPMI 1640 (Sigma) medium. Cells were counted and resuspended in 164 ml of fresh medium contained in a 150 cm(^2) cell culture flask (ventilated lid). The flasks were incubated at 37°C in a 5% CO(_2) atmosphere for 30 min to allow CO(_2) balance. 26 ml of cells were placed in a universal tube, the lid was tightly closed to maintain the balance of CO(_2) in the medium and the tube was then transferred to an RF exposure facility and placed in a 37 °C incubator. 12 ml of cells were placed in a petri dish of the RF resonance cavity and 12 ml of cells were placed in the second resonance cavity and exposed. The cell density is 3-8 (times ) 10(^6) cells and cells are used in sections 8-12. Cells were exposed to RF or sham for 6 or 18 min and various duty cycles were assayed. Post-exposure, the cells were placed in a universal tube for transport back to the laboratory. The samples were then transferred to petri dishes and incubated at 37 °C in 5% CO(_2) atmosphere for 3 hours. Cell viability (as described below) was determined immediately after this 3 h incubation.
12 ml cell samples exposed to RF/sham were centrifuged (300 (times ) g; 5 min; 5 °C), washed with ice cold phosphate buffered saline (PBS) and stained with annexin V-FITC and propidium iodide (PI). ) using the Immunotech Annexin V-FITC kit (Beckman Coulter) according to the manufacturer’s instructions. The prepared samples were analyzed using a BD FACSCanto II flow cytometer with FACsDiva version 6 software, to show viable cell populations (unstained), apoptotic cells (annexin V-FITC stained) and necrotic cells (PI and Annexin V-FITC stained).
Results and analysis
Temperature measurements were carried out with the Opsens fiber optic thermometer system which consisted of a fiber optic probe and a measurement system. Temperature was measured before and after RF exposure, but it was not possible to record during exposure because the resonant cavity is a closed system. Before or immediately after exposure, the probe is placed into the medium, the resonant cavity cover is then replaced, and this holds the probe in place. The incubator door is closed and temperature changes are recorded by the fiber optic unit. For post-exposure cooling measurements, exposure time ranges and duty cycles are used.
To provide an indication of the temperature of the media in the petri dish during RF exposure, a 5 point temperature indication disc (Part Number: SC5 / Type 01 / C) manufactured by Omega (Manchester, UK) was used. An indication disc is attached to the center of the petri dish at a fixed location for each exposure; various duty cycles and duration of exposure were investigated. Each circle on the disc indicates a specific temperature and, if this temperature is reached during exposure, the disc changes color permanently.
Cell viability measurements were carried out using the Annexin V-FITC and PI methods at 3 hours after the RF/Sham exposure method 6 or 18 minutes and the results of 3 independent experiments at each exposure duration are shown in Figure 13. Experiments were carried out using a constant peak field and increasing duty cycle (by reducing the interpulse interval). With increasing exposure time, the interpulse interval which affects the viability also increases. During an exposure duration of 6 min, an interpulse interval of 5.5 s or less resulted in decreased cell viability, in the absence of viable cells when the interpulse interval was 2.75 s. For a longer 18 min exposure duration, reduced cell viability was observed at longer interpulse intervals, 11 s or less. No change in viability was observed at 11 s (6 min duration) or 55 s (18 min duration) interpulse intervals. The results also showed that the change in viability was caused by necrosis rather than apoptosis.
Effect of 6/18 min exposure to interpulse intervals of 2.75, 4.4, 5.5, 11, 55 and 110 s (RF 1.2 GHz, 550 ns pulses) on cell viability (data shown represent mean (+ /-) SEM ( n=3)); (a) % live cells; (b) % necrotic cells; (c) % of apoptotic cells.
The ultimate aim of this work is to investigate the possible relationship between peak field intensity and biological effects. To do this, it is first necessary to determine the threshold for the heating-induced effect. The temperature measurement of the culture medium was carried out by placing the optical fiber sensor (n = 3) into the petri dish as soon as possible (approximately 10 seconds) after exposure. Figure 14 shows representative temperature recordings after 18 min exposure at interpulse intervals of 2.75, 5.5, and 11 s. Temperatures greater than 60 °C were achieved with an interpulse interval of 2.75 s. The peak temperatures recorded were recorded at 65°C, 55°C, and 42.5°C, respectively, followed by cooling curves. Increasing the interpulse interval resulted in less heating, and no detectable temperature rise for the 55 s interpulse interval (data not shown).
Measurement of media temperature immediately after 18 min exposure, at interpulse intervals of 2.75, 5.5 and 11 s.
Independent indication of temperature for 18 minutes of exposure is achieved using a temperature indicator disc. The results of 3 independent experiments at an exposure duration of 18 min are shown in Figure 15. It was observed that the 11 s interpulse interval raised the indoor temperature by at least 46 °C (but lower than 50 °C). Interpulse intervals of 2.75 and 5.5 s increase the temperature above 54 °C while the interpulse intervals of 55 and 110 s do not increase the temperature in the room above 40 °C.
The temperature indicator disk response to an interpulse interval of 18 minutes (RF 1.2 GHz, pulse 550 ns), (n = 3).
Based on the observations from Figure 13, it can be concluded that a temperature range of 43–46 °C is required to initiate a decrease in cell viability in the resonance cavity space. The cooling curve shown in Figure 14 and the temperature indication disk data given in Figure 15 further strengthen this understanding.
Discussion
Additional work was performed on temperature indicator discs for 18 min of exposure duration. The 11 s interpulse interval raised the ambient temperature to 46 °C, however, the 55 s interpulse interval did not increase the ambient temperature above 40 °C. Typical in vitro heat shock exposure involves heating mammalian cells to 42-45 °C for 20-60 minutes and then returning them to a normothermic temperature (37 °C)58. Exposure of 18 min to an 11 s interpulse interval produced conditions similar to heat shock exposure and resulted in cell viability of less than 40% (Fig. 13).
Exposing cells to RF for 18 min (Fig. 13) showed that cell viability began to decrease at interpulse intervals less than 55 s. These results indicate that the heating threshold is between 55 and 11 s interpulse interval. Temperatures measured with temperature indicator discs during RF exposure with interpulse intervals of 55 and 11 s (Fig. 15) reached 40 °C and 46 °C, respectively. Previous work by the team showed that human T cells heated to 43 °C were damaged but mechanisms were upregulated to limit and/or reverse the damage. However, upon heating to 46 °C, the cells appeared to have accumulated too much damage and cell death occurred in much greater numbers59,60. Previous studies support our current findings. To further investigate the heating threshold, initial experiments showed that an interpulse interval of 27.5 s during 18 min exposure resulted in no viable cells, and an interpulse interval of 41.25 s resulted in 23% cell viability after exposure. Further work will result in complete data sets for the interpulse intervals of 27.5 and 41.25 s, including completing cell viability studies and assessing the temperature in the RF exposure chamber during exposure with temperature indicator discs. The results of this study indicated that the threshold for heating-induced cell death was between the 41.25 and 55 s interpulse intervals, with a constant peak field and 18 min exposure.
The resonant cavity system has been used previously for applications such as in vivo dental dosimetry44, homogeneous enzymatic hydrolysis of sucrose45, measurement of general solvent dielectrics46 and for electron paramagnetic resonance spectroscopy47. They have also been used to expose biological preparations to RF33,39,40,41,42 fields. The cavity described in this study compares favorably with this published system, with a Q factor of 5920 under no-load conditions, compared to 3000 in40, 6500 in44, and 2073 in47. With an input power of 200 W, the bay is capable of achieving a uniform electric field strength of 125 kV/m under no-load conditions, higher than reported for some other systems, e.g. 2 kV/m41.45 or 10 kV/m33. When filled with petri dishes containing cell culture media, even though the Q factor dropped to 57, the cavity was able to generate a field strength of 63 kV/m, which was better than the 30 kV/m reported by Hamzah et al. in a cavity filled with a sample of methanol46.
Experiments with cell cultures showed that these cavities were capable of causing changes in cell viability associated with heating to 46 °C. Cell death induced by necrosis was observed at lower duty cycles for longer duration exposures, consistent with that due to temperature rise. This was confirmed by temperature measurements immediately after exposure and by a temperature indicator disc attached to the petri dish during exposure. This shows good agreement and suggests that an ambient temperature of 46 °C is required to induce cell death under these exposure conditions.
Although there is a delay between the end of exposure and the start of temperature recording with the fiber-optic probe, the recording shows a smooth cooling curve that allows for estimating the return to the end of the exposure time. Temperature indicator discs have a much coarser resolution (46 °C) but, because they can be included in the exposure, they provide useful confirmation for Opsens measurements.
These experiments have characterized the exposure conditions and temperature thresholds for heating-induced cell death, and have shown that this effect can be avoided by reducing the duty cycle (increasing the interpulse interval) while maintaining the same peak electric field strength in the pulses. Future studies will utilize longer interpulse intervals to look for effects associated with peak field strength rather than heating due to energy absorption. To do this, it is necessary to distinguish temperature-dependent non-lethal effects. There may be a change in gene expression, for example, at a lower level of heating that is compensated by the cell and does not result in cell death.
Conclusion
This study used the human lymphoid cell line (GG0257) for ease of culture and consistency between plates; however, primary cell cultures might be expected to be more susceptible to environmental stressors and these will be used in future experiments to look for peak field effects. Comparisons between cell lines and primary cells may also be useful.
Data availability
Although, the disassembled cavity is capable of achieving a field strength greater than the recommended exposure limit of 100 kV/m in the IEEE 2005 standard, under load conditions the maximum electric field strength drops to 63 kV/m. Taking into account the external field coupling losses into the human body, however, a field strength of 63 kV/m in cell culture most likely represents what would be achieved in many tissues with much higher external field intensities.
References
A detailed study of a resonance-based cavity system for the exposure of human cells to very high electric fields has been presented. The exposure system was numerically designed and experimentally tested in both no-load and load configurations with an effective impedance matching mechanism to reduce detuning and impedance mismatches caused by loading of human cell cultures in petri dishes. This system offers good tunability, high level electric field, and uniformity of SAR across cell culture media. The cavities are also gold plated for enhanced performance. Good agreement between simulation and reflection coefficient measurements has been demonstrated by the system operating in the 1000–2000 MHz range and 200 W input. Detailed measurements on cell GG0257 using the exposure system were then carried out to verify the working of the developed system. This study also investigated the effect of RF on cell viability and has provided valuable information to allow a pulse repetition rate to be selected that is low enough to avoid heat-induced changes. The results indicate that the designed system is potentially a good candidate for analyzing cell culture changes at the recommended exposure limit of 100 kV/m.
Data generated or analyzed during this research is available from the relevant authors upon reasonable request.
International Commission on Non-Ionizing Radiation Protection (ICNIRP): Guidelines for limiting exposure to time-varying electric, magnetic and electromagnetic fields (up to 300GHz). Health Physics 118(5), 483–524 (2020).
IEEE International Committee on Electromagnetic Safety. IEEE standard for safety levels with respect to human exposure to radio frequency electromagnetic fields, 3kHz to 300GHz. IEEE Std C95 (1–2005), 1-238 (2005).
Poljak, D. & Cvetkovic, M. Human Interaction with Electromagnetic Fields (Academic Press, Cambridge, 2019).
MATHEMATICS
Google scholarship
IEEE Standards Coordinating Committee on Non-Ionizing Radiation Hazards. Standard for safety levels with respect to human exposure to radio frequency electromagnetic fields, 3kHz–300 GHz). IEEE Std C95 (1-1999), 1–83 (1999).
International Commission for the Protection of Non-Ionizing Radiation (ICNIRP): Guidelines for limiting exposure to time-varying electric, magnetic and electromagnetic fields (up to 300 GHz). Health Physics 74(4), 494–522 (1998).
Tattersall, J. E. H. et al. Effect of a low-intensity radiofrequency electromagnetic field on electrical activity in rat hippocampal slices. Brain Res. 904(1), 43–53 (2001).
CAS
PubMed
Google scholarship
Alfadhl, Y., Wang, Z., Chen, X., Tattersall, J.H., & Holden, S. J. Numerical dosimetry of the thin rat hippocampus in a controlled rf exposure system. URSI (2002).
Ardoino, L., Lopresto, V., Mancini, S., Pinto, R. & Lovisolo, G. A. 1800 MHz in vitro exposure device for experimental study of the effects of mobile communication systems. radiation. Protective Dosimetry 112(3), 419–28 (2004).
CAS
Google scholarship
Liberti, M., Apollonio, F., Paffi, A., Pellegrino, M. & D’Inzeo, G. Coplanar-waveguide system for cell exposure during electrophysiological recording. IEEE Trans. Microwave Theory Technology. 52(11), 2521–2528 (2004).
ADVERTISEMENT
Google scholarship
Leveque, P., Dale, C., Veyret, B. & Wiart, J. Dosimetric analysis of a 900 MHz mouse head exposure system. IEEE Trans. Microwave Theory Technology. 52(8), 2076–2083 (2004).
ADVERTISEMENT
Google scholarship
Paffi, A., Liberti, M., Lopresto, V., Merla, C., Lodato, R., Lovisolo, G. A., & Apollonio, F. A wire patch cell exposure system for in vitro experiments at wi-fi frequencies. IEEE Trans. Microwave Theory Technology. 58 (12 PART 2), 4086–4093 (2010).
Whiting, R., Wright, A., Lindsay, C., Scott, I., Ur-Rehman, M., Alfadhl, Y., Chen, X., & Tattersall, J. Effect of pulsed radio frequency fields on cell culture viability. BioEM (2014).
Ur-Rehman, M., Alfadhl, Y., Chen, X., Tattersall, J., & Scott, I. W. K. Designing and studying a very high electric field exposure system for exposure to human cells. BioEM (2014).
Lameth, J. et al. Acute neuroinflammation enhances cell response to 1800MHz GSM electromagnetic fields in the rat cerebral cortex. Neurotoxicity Res. 32(3), 444–459 (2017).
Occelli, F. et al. Single exposure to GSM-1800 MHz signals during an acute neuroinflammatory reaction can alter neural responses and microglial morphology in the primary auditory cortex of mice. Neuroscience 385, 11-24 (2018).
CAS
PubMed
Google scholarship
Zhao, J. An extrapolation method for efficient and accurate numerical dosimetry of resonant exposure setups. IEEE Trans. Microwave Theory Technology. 66(2), 774–783 (2018).
ADVERTISEMENT
Google scholarship
Ouadah, N.S., Blazy, K. & Villegier, A.-S. Effect of radio frequency electromagnetic fields on thermal sensitivity in mice. int. J.Environment. res. Public Health 17 (20), 7563 (2020).
CAS
PubMed Center
Google scholarship
Lameth, J. et al. Effects of single-head exposure to GSM-1800 MHz signals on transcriptome profiles in the rat cerebral cortex: Enhanced gene response under proinflammatory conditions. Neurotoxicity Research 38(1), 105–123 (2020).
CAS
PubMed
PubMed Center
Google scholarship
Hansen, J.W. et al. Systematic review of in vitro and in vivo radiofrequency exposure methods. IEEE Rev. biomed. eng. 13, 340–351 (2020).
PubMed
Google scholarship
Crawford, M. L. Generation of standard EM fields using TEM transmission cells. IEEE Trans. electromagnetic. compatible. EMC–16(4), 189–195 (1974).
Guy, A.W., Chou, C.K. & McDougall, J. A. A quarter century of in vitro research: a new look at exposure methods. Bioelectromagnetics 20(S4), 21–39 (1999).
Nikoloski, N., Frohlich, J., Samaras, T., Schuderer, J. & Kuster, N. Re-evaluation and improved design of the in vitro exposure unit of TEM cells for replication studies. Bioelectromagnetics 26(3), 215-224 (2005).
PubMed
Google scholarship
Kumar, G. et al. Evaluation of the effects of the hematopoietic system after exposure to radiofrequency radiation in vitro in rats. int. J. Radiation. violin. 87(2), 231–240 (2011).
CAS
PubMed
Google scholarship
Schnborn, F., Pokovic, K., Wobus, A. & Kuster, N. Design, optimization, realization, and analysis of an in vitro system for exposure of embryonic stem cells at 1.71 GHz. IEEE Trans. biome. eng. 21, 372–384 (2000).
Hagan, T., Chatterjee, I., McPherson, D. & Craviso, G. L. A new waveguide-based radio frequency/microwave exposure system for studying nonthermal effects on neurotransmitter release-to-difference time domain modeling. IEEE Trans. Plasma Science. 32(4), 1668–1676 (2004).
ADVERTISEMENT
Google scholarship
Takashima, Y. et al. Effects of continuous and intermittent exposure to RF fields with various SARs on cell growth, survival, and cell cycle distribution. Bioelectromagnetics 27(5), 392–400 (2006).
PubMed
Google scholarship
De Prisco, G., d’Ambrosio, G., Calabrese, M. L., Massa, R. & Juutilainen, J. SAR and evaluation of the efficiency of the 900 MHz waveguide chamber for cell exposure. Bioelectromagnetics 29(6), 429–438 (2008).
PubMed
Google scholarship
Varela, J.E., Page, J.E. & Esteban, J. Design, implementation, and dosimetry analysis of an s-band waveguide in vitro system for exposing cell culture samples to a pulsed field. Bioelectromagnetics 31, 479–487 (2010).
PubMed
Google scholarship
Sefidbakht, Y. et al. Effect of 940MHz EMF on luciferase solutions: structure, function, and dielectric studies. Bioelectromagnetics 34(6), 489–498 (2013).
PubMed
Google scholarship
Houston, B. J., Nixon, B., King, B. V., Aitken, R. J. & De Iuliis, G. N. Investigating the origin of 1800 MHz radio frequency electromagnetic radiation that causes damage to germ cells and rat spermatozoa immortalized in vitro. Frontiers in Public Health 6, 270 (2018).
PubMed
PubMed Center
Google scholarship
Funding
Zhao, J. Using oblique waveguide cavities to increase resonance exposure in cultured cells. IEEE Trans. micro. Theory Technology. 66(5), 2522–2534 (2018).
Author information
Affiliations
ADVERTISEMENT
Google scholarship
Schnborn, F., Pokovic, K. & Kuster, N. Basis for optimization of in vitro exposure equipment for health evaluation of mobile communications. Bioelectromagnetics 22(8), 547–559 (2001).
Schuderer, J., Samaras, T., Oesch, W., Spitting, D. & Kuster, N. High-peak SAR exposure unit with severe exposure and environmental control for in vitro experiments at 1800MHz. IEEE Trans. micro. Theory Technology. 52(8), 2057–2066 (2004).
Contributions
ADVERTISEMENT
Ethics declarations
Competing interests
Google scholarship
Additional information
Publisher’s note
Schuderer, J., Spat, D., Samaras, T., Oesch, W. & Kuster, N. In vitro exposure system for RF exposure at 900 MHz. IEEE Trans. micro. Theory Technology. 52(8), 2067–2075 (2004).
Rights and permissions
ADVERTISEMENT
About this article
Cite this article
Google scholarship
Comments
Gerber, H. L. et al. Analytical and experimental dosimetry of cell cultures in T-25 flasks placed in a thermally controlled waveguide. IEEE Trans. Plasma Science. 34(4), 1449–1454 (2006).

